Our Science
The Theriva™ Difference
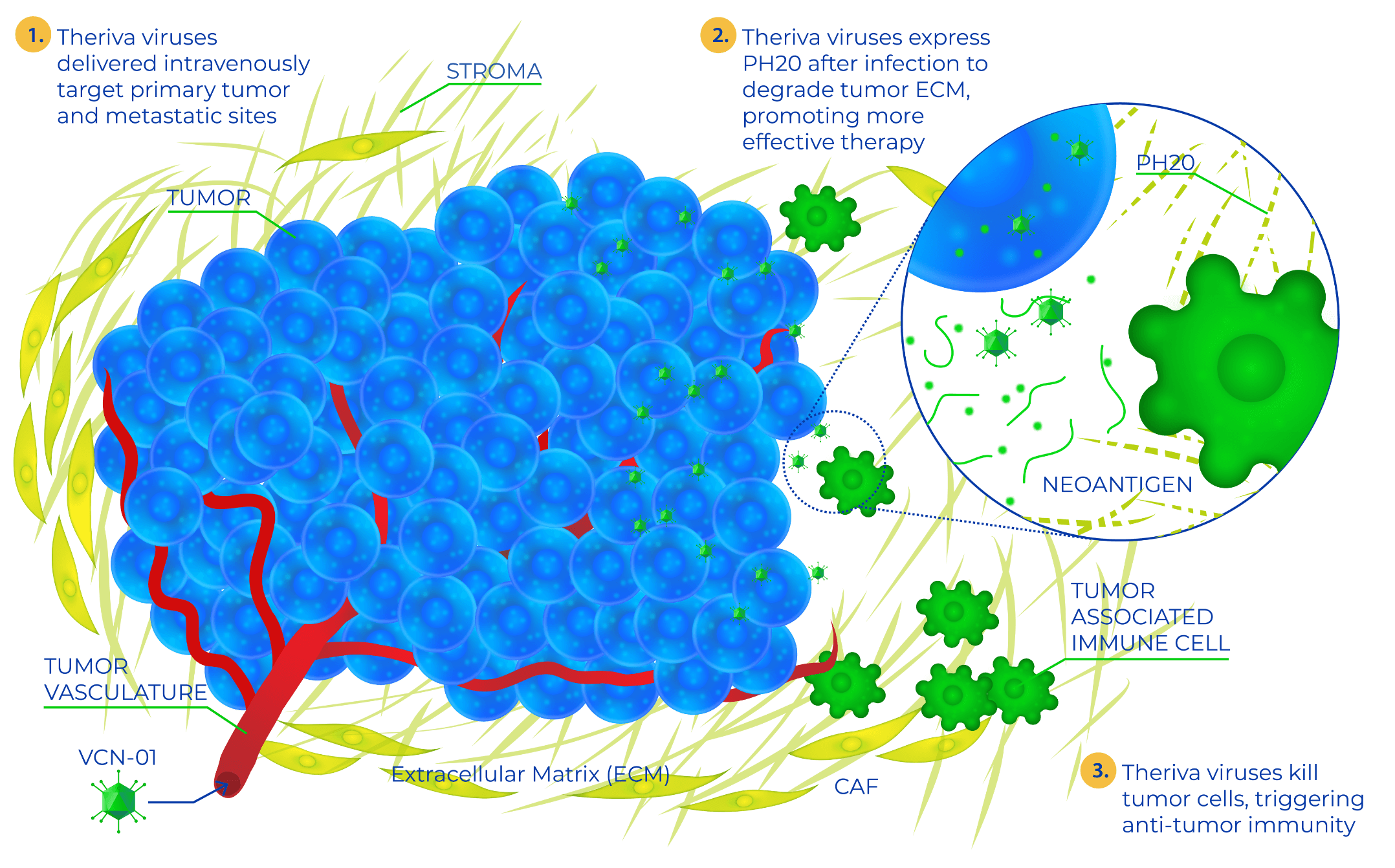
Theriva is developing highly differentiated oncolytic adenoviruses that are designed to overcome the challenges of accessibility, availability, and administration and can potentially enable multiple dosing to improve efficacy.
The unique therapeutic properties of Theriva’s oncolytic viruses represent a new class of cancer treatments that are designed to selectively destroy tumor cells and enhance the effects of current standard-of-care therapies. Engineered for systemic delivery, Theriva’s oncolytic viruses can be administered intravenously or as a direct injection into the tumor or tumor compartment (intratumoral, intravitreal).
To improve efficiency, Theriva’s oncolytic viruses are designed for:
Systemic DELIVERY
Eliminates the need for invasive surgeries and permits the oncolytic virus to reach both the target tumor and metastases throughout the body
Selective REPLICATION
in tumor cells, which minimizes toxicity and destroys the tumor while avoiding damage to healthy tissues
Stromal degradation
Intended to break down the tumor’s protective barrier, improve tumor access by the oncolytic virus and other cancer therapies, and expose the tumor to robust and persistent attack by the patient’s immune system
This unique combination of features means that Theriva’s oncolytic viruses are particularly well-positioned for concomitant administration with other cancer treatment modalities (chemotherapy, immunotherapy, etc.) and may enhance the efficacy of both standard-of-care and developmental cancer therapies.
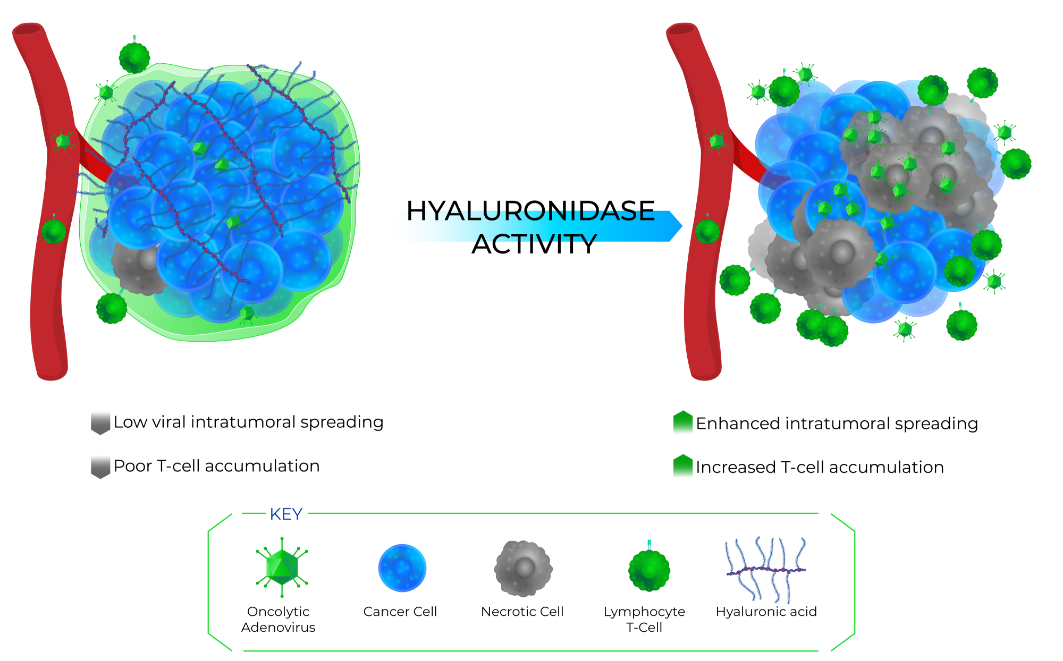
Hyaluronidase Technology
Solid tumors are complex organ-like structures consisting of cancer cells, vasculature, and tumor matrix. This dense extracellular matrix, also known as stroma, hampers the intratumoral accessibility of chemotherapy, antitumor immune cells, and oncolytic viruses. Our viruses are “armed” with a soluble version of human sperm hyaluronidase (hyal) PH20 to enhance virus spreading and help the actions of chemotherapy and the immune system.
VCN-01 (zabilugene almadenorepvec): Lead Candidate
Theriva’s leading systemic, selective, and stroma-degrading oncolytic virus clinical candidate is VCN-01 (zabilugene almadenorepvec), which successfully completed a phase 2 clinical trial in patients with pancreatic ductal adenocarcinoma (PDAC) and has been evaluated in investigator sponsored studies in a number of indications.
VCN-01 is characterized by 4 interconnected evolutions in oncolytic virus design that enable intravenous delivery at higher doses than have been feasible for other oncolytic viruses:
High replication rate
Increases manufacturing yields and is expected to enable an antitumor effect even if low levels of the oncolytic virus are delivered to the tumor
Tumor-selective replication
Intended to maximize antitumor effects, minimize potential off-target effects, and limit damage to healthy tissues
Expression of hyaluronidase (PH20)
Degrades tumor stromal hyaluronic acid, a substance that has been strongly associated with reduced tumor immunogenicity and poor prognosis in patients with PDAC and ovarian cancer
Liver detargeting
Intended to reduce the loss of VCN-01 through hepatic clearance and avoids potential liver toxicities
VCN-01 has been uniquely engineered to ensure that PH20 expression occurs only after VCN-01 has entered the tumor and undergone a viral replication cycle. This feature ensures a tumor-focused release of PH20 and provides a powerful biomarker for measuring VCN-01 tumor delivery and activity.
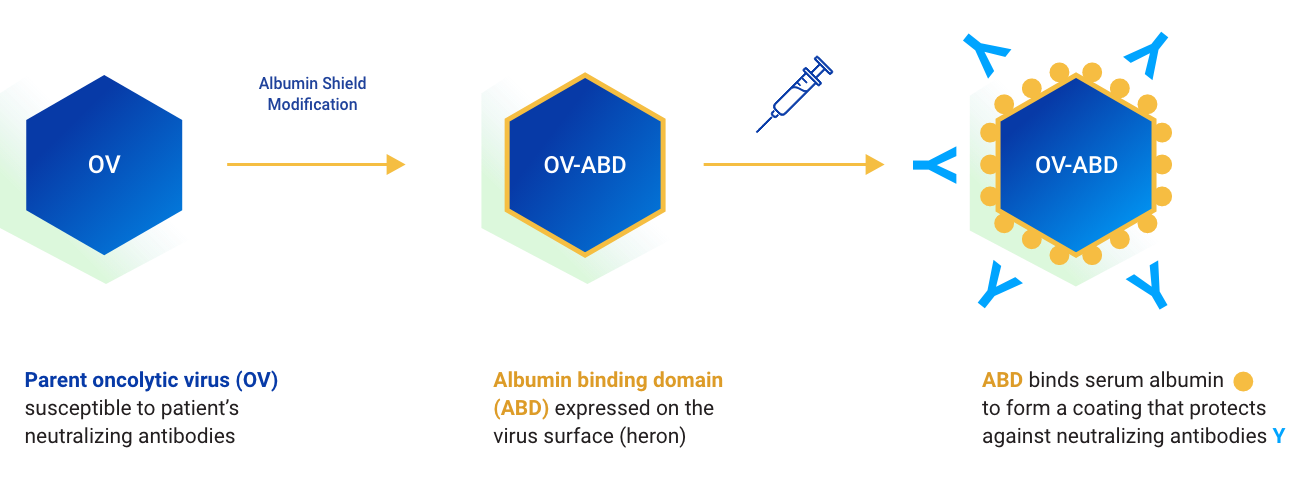
Albumin Shield™ Technology
Building on the clinical advancement of VCN-01, Theriva developed the Albumin Shield™ technology to protect oncolytic viruses from circulating anti-oncolytic virus antibodies after systemic administration. We anticipate that this technology will enable multiple oncolytic virus doses to be administered in therapeutic cycles to treat particularly refractory tumors.
Preclinical proof of concept for the Albumin Shield™ technology has been demonstrated with the archetype VCN-11, and a range of Albumin Shield™ oncolytic viruses with different therapeutic payloads are currently under consideration. Other technologies in earlier stages of development are also being explored by our experienced scientific team.

Clinical Evidence to Date
To date, more than 140 patients have been dosed with VCN-01 in phase 1 and phase 2 clinical trials in patients with a broad range of cancers, including PDAC, retinoblastoma, colorectal cancer, head and neck squamous cell carcinoma (HNSCC), brain cancers, and ovarian cancers.
VCN-01 has been well tolerated in clinical trials and accumulated clinical data support the proposed VCN-01 mechanisms of action. In the VIRAGE randomized, controlled Phase 2b trial, metastatic pancreatic cancer patients treated first-line with intravenous VCN-01 plus gemcitabine/nab-paclitaxel standard of care (SoC) chemotherapy demonstrated increased overall survival, progression free survival and duration of response compared to patients treated with SoC chemotherapy alone. Greater improvements in survival were observed in patients treated with two VCN-01 doses and the survival profiles supported a VCN-01 mediated antitumor immune response.
We have regulatory alignment with the FDA and EMA on the design of a proposed Phase 3 clinical trial of VCN-01 in metastatic pancreatic cancer and we are planning a Phase 2/3 clinical trial in retinoblastoma.